Hits:
DOI number:10.1021/jacs.5c22595
Journal:Journal of the American Chemical Society
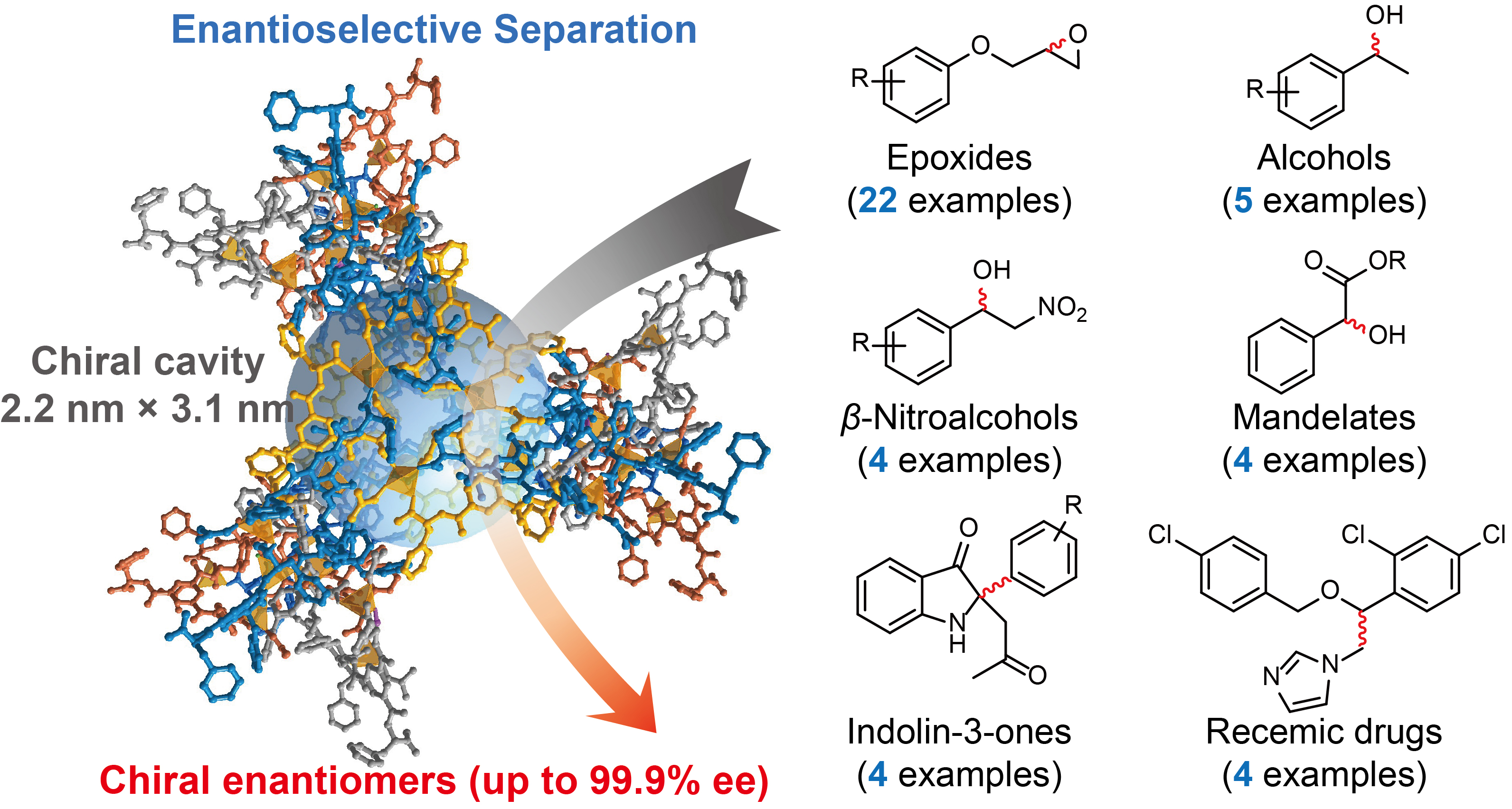
Abstract:The selective separation of enantiomers is critical in pharmaceutical production, while conventional chiral sorbents always suffer from the trade-off between selectivity and the substrate scope. Herein, inspired by a natural unspecific peroxygenase with large protein channels, we developed a homochiral metal–organic framework (MOF) constructed from flexible phenylalanine-derived ligands and zinc ions. This MOF features a giant chiral cavity with a size of 2.2 × 3.1 nm, decorated with 42 chiral phenylalanine residues, which serves as a solid sorbent for the highly enantioselective adsorption and separation of diverse chiral compounds, including aromatic epoxides, β-nitroalcohols, mandelate derivatives, secondary alcohols, indolin-3-ones, α-methylbenzylamine, and limonene. Most importantly, benefiting from its large pores, the MOF demonstrates versatile utility in resolving the enantiomers of extraordinarily bulky substrates and structurally complex chiral pharmaceuticals, which can be further processed into a polymer matrix for membrane separation, enabling an integrated, chromatography-free route from batch-scale adsorption and separation. This material can be readily recovered and reused without an apparent loss of performance. Adsorption experiments and theoretical calculations reveal that the chiral recognition and separation originate from the distinct binding affinity of enantiomers within the MOF’s chiral pore environment, presenting a scalable platform for process-intensified chiral separations.
Indexed by:Journal paper
Correspondence Author:Zhu chengfeng* et al.
Discipline:Natural Science
Document Type:J
Translation or Not:no
Date of Publication:2026-03-12
Included Journals:SCI
Links to published journals:https://pubs.acs.org/doi/10.1021/jacs.5c22595


