Hits:
DOI number:10.1021/jacs.1c03652
Journal:Journal of the American Chemical Society
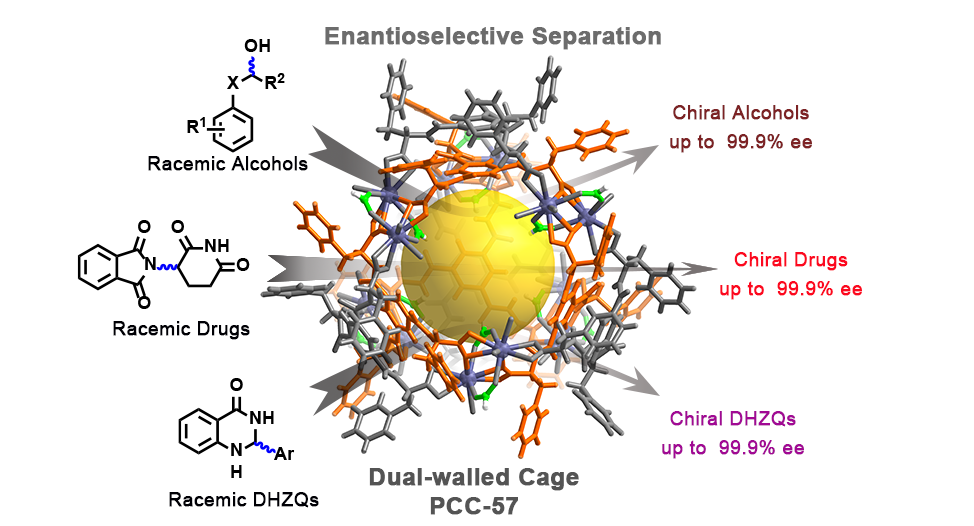
Abstract:It is extremely difficult to anticipate the structure and the stereochemistry of a complex, particularly when the ligand is flexible and the metal node adopts diverse coordination numbers. When trivalent lanthanides (LnIII) and enantiopure amino acid ligands are utilized as building blocks, self-assembly sometimes yields rare chiral polynuclear structures. In this study, an enantiopure carboxyl-functionalized amino acid-based ligand with C3 symmetry reacts with lanthanum cations to give a homochiral porous coordination cage, (Δ/Λ)12-PCC-57.The dodecanuclear lanthanide cage has an unprecedented octahedral “cagein-cage” framework. During the self-assembly, the chirality is transferred from the enantiopure ligand and fixed by the binuclear lanthanide cluster to give 12 metal centers that have either Δ or Λ homochiral stereochemistry. The cage exhibits excellent enantioselective separation of racemic alcohols, 2,3-dihydroquinazolinones, and multiple commercially available drugs. This finding exhibits a rare example of a multinuclear lanthanide complex with a dual-walled topology and homochirality. The highly ordered self-assembly and self-sorting of flexible amino acids and lanthanides shed light on the chiral transformation between different complicated artificial systems that mimic natural enzymes.
Indexed by:Journal paper
Correspondence Author:Zhu Chengfeng* et al.,
Discipline:Natural Science
Document Type:J
Volume:143
Issue:32
Page Number:12560-12566
Translation or Not:no
Date of Publication:2021-08-03
Included Journals:SCI
Links to published journals:https://pubs.acs.org/doi/10.1021/jacs.1c03652


